Spawning Activities, Early Development, and Larvae Growth of the Arfak Rainbowfish in Laboratory Condition: A Preliminary Study for Its Breeding Captivity
DOI:
https://doi.org/10.46252/jsai-fpik-unipa.2021.Vol.5.No.3.165Keywords:
endemic fish, Melanotaenia, Captive breedingAbstract
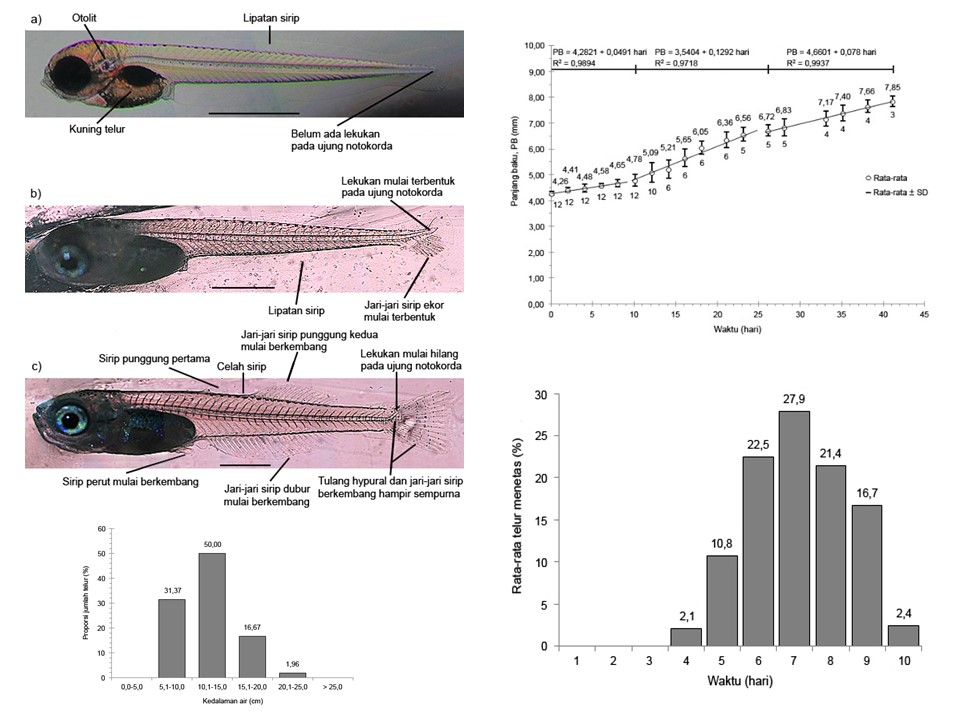
Comprehensive information regarding the reproduction, early development, and growth of larvae is very important in the efforts of captive breeding and reintroduction of endangered fish species. One of the species that is starting to be threatened is the Arfak rainbowfish, Melanotaenia arfakensis which is already in the vulnerable category and its information is still relatively limited. Therefore, this study was conducted to describe the spawning activity, early development, and growth of the larvae of this rainbow fish species under laboratory conditions. Experiments on a laboratory scale were carried out in March-September 2017. The fish used came from the Nimbai Stream. A total of three pairs of individual males and females ranging in size from 46.5 to 60.1 mm were selected for treatment. However, only one pair was successfully observed spawning activity completely. Spawning takes place between morning and noon in three spawning periods. In each period, spawning lasts between 8 and 11 days. Between spawning periods takes 14 to 22 days. After fertilization, the eggs are attached with filaments to the spawning substrate and placed at a depth of 7.3-24.3 cm from the surface of the water. One group of eggs spawned consists of 78-116 eggs with a diameter range between 1.05 and 136 mm. The eggs hatch in a period of 4 to 10 days. The body length of the newly hatched larvae ranges from 4.13-4.40 mm and will reach a size of 7.85 mm with a survival rate of 48.1% after 41 days. The results of this study found several advantages from the characteristics of spawning and growth in the early stages that are useful for captive breeding efforts and reintroduction of this rainbowfish to their natural habitat. Thus, its population in the Prafi River system can be maintained.
Downloads
References
Balon EK. (1984). Patterns in the evolution of reproductive styles in fishes. In: Potts GW, Wootton RJ. (eds.). Fish reproduction: strategies and tactics. New York (USA): Academic Press. pp. 35-52
Brown C. (2003). Habitat-predator association and avoidance in rainbowfish (Melanotaenia spp.). Ecology of Freshwater Fish. 12(2): 118-126
Chivers DP, Smith RJF. (1994). Flathead minnows (Pimephales promelas) learn to recognize chemical stumuli from high-risk habitats by the presence of alarm substance. Behavioural Ecology. 6(2): 155-158
Chivers DP, Smith RJF. (1995). Chemical recognition of risky habitats is culturally transmitted among flathead minnows, Pimephales promelas (Osteichthyes, Cyprinidae). Ethology. 99(4): 286-296
Crowley LELM, Ivantsoff W. (1982). Reproduction and early stages of development in two species of Australian rainbowfishes, Melanotaenia nigrans (Richardson) and Melanotaenia splendida inornata (Castelnau). Australian Zoologist. 21(1): 85-95
Crowley LELM, Ivantsoff W, Allen GR. (1986). Taxonomic position of two crimson-spotted rainbowfish, Melanotaenia duboulayi and Melanotaenia fluviatilis (Pisces: Melanotaeniidae), from eastern Australia, with special reference to their early life-history stages. Australian Journal of Marine and Freshwater Research. 37(3): 385-398
Doupe RG, Schaffer J, Knott MJ, Burrows DW. (2009). How might an exotic fish disrupt spawning success in a sympatric native species? Marine and Freshwater Research. 60(5): 379-383
Einum S, Fleming IA. (1999). Maternal effects of egg size in brown trout (Salmo trutta): norms of reaction to environmental quality. Proceedings of the Royal Society B. 266(1433): 2095-2100
Einum S, Fleming IA. (2000). Highly fecund mothers sacrifice offspring survival to maximize fitness. Nature. 405: 565-567
Einum S, Fleming IA. (2004). Environmental unpredictability and offspring size: Conservative versus diversified bet-hedging. Evolutionary Ecology Research. 6: 443-455
Flint N, Pearson RG, Crossland MR. (2018). Reproduction and embryo viability of a range-limited tropical freshwater fish exposed to fluctuating hypoxia. Marine and Freshwater Research. 69(2): 267-276
Greig SM, Sear DA, Carling PA. (2005a). The impact of fine sediment accumulation on the survival of incubating salmon progeny: implications for sediment management. Science of The Total Environment. 344(1-3): 241-258
Greig SM, Sear DA, Smallman D, Carling PA. (2005b). Impact of clay particles on cutaneous exchange of oxygen across the chorion of Atlantic salmon eggs. Journal of Fish Biology. 66(6): 1681-1691
Greig SM, Sear DA, Carling PA. (2007). A field-based assessment of oxygen supply to incubating Atlantic salmon (Salmo salar) embryos. Hydrological Processes. 21(22): 3087-3100
Hattori A, Warburton K. (2003). Microhabitat use by the rainbowfish Melanotaenia duboulayi in a subtropical Australian stream. Journal of Ethology. 21(1): 15-22
Hismayasari IB, Rahayu S, Marhendra APW. (2015). Ovary maturation stages histology and follicles diameter of Melanotaenia boesemani rainbowfish ovary from district of North Ayamaru, Maybrat Regency, West Papua. Journal of Morphological Science. 32(3): 157-164
Houde ED. (1994). Differences between marine and freshwater fish larvae: implications for recruitment. ICES Journal of Marine Science. 51(1): 91-97
Humphrey C, Klumpp DW, Pearson R. (2003). Early development and growth of the eastern rainbowfish, Melanotaenia splendida splendi-da (Peters) I. Morphogenesis and ontogeny. Marine and Freshwater Research. 54(1): 17-25
Humphries P, King AJ, Koehn JD. (1999). Fish, flows and flood plains: links between freshwater fishes and their environment in the Murray-Darling River Systems, Australia. Environmental Biology of Fishes. 56(1-2): 129-151
Hunter JR. (1984). Feeding ecology and predation of marine fish larvae. In: R. Lasker (ed.). Marine fish larvae: morphology, ecology and relation to fisheries. Seattle, WA (USA): Washington Sea Grant Program. pp. 34-77
Hutchings JA. (1997). Life history responses to environmental variability in early life. In: Chambers RC, Trippel EA. (eds.). Early life history and recruitment in fish populations. Fish and Fisheries Series 21. London (UK): Chapman and Hall. pp. 139-168
IUCN. (2018). The IUCN red list of threatened species. Version 2017-3. [accessed: 7 May 2018]. http://www.iucnredlist.org
Kadarusman, Sudarto, Paradis E, Pouyaud L. (2010). Description of Melanotaenia fasinensis, a new species of rainbowfish (Melanotaeniidae) from West Papua, Indonesia with comments on the rediscovery of M. ajamaruensis and the endagered status of M. parva. Cybium. 34(2): 207-215
Kendall AW Jr, Ahlstrom EH, Moser HG. (1984). Early life history stages of fishes and their characters. In: Moser HG, Richards WJ, Cohen DM, Fahay MP, Kendall AW Jr, Richardson SL (eds.). Ontogeny and systematics of fishes. California (USA): American Society of Ichthyologists and Herpetologists. pp 11-22
Kjesbu OS, Witthames PR, Solemdal P, Walker MG. (1998). Temporal variations in the fecundity of Arcto-Norwegian cod (Gadus morhua) in response to natural changes in food and temperature. Journal of Sea Research. 40(3-4): 303-321
Leatemia SPO, Manangkalangi E, Lefaan PT, Peday HFZ, Sembel L. (2017). Makroavertebrata bentos sebagai bioindikator kualitas air Sungai Nimbai Manokwari, Papua Barat. Jurnal Ilmu Pertanian Indonesia. 22(1): 25-33
Lefaan PT, Peday HFZ, Leatemia SPO, Sembel L, Manangkalangi E. (2019). Struktur vegetasi riparia dan implikasinya terhadap kondisi habitat ikan pelangi arfak, Melanotaenia arfakensis di Sungai Nimbai, Manokwari Papua Barat. Samakia: Jurnal Ilmu Perikanan. 10(1): 38-56
Leu M-Y, Meng P-J, Huang C-S, Tew KS, Kuo J, Liou C-H. (2010). Spawning behavior, early development and first feeding of the bluestriped angelfish [Chaetodontoplus septentrionalis (Temminck & Schlegel, 1844)] in captivity. Aquaculture Research. 41: e39-e52
Lobon-Cervia J, Utrilla CG, Rincøn PA, Amezcua F. (1997). Environmentally induced spatio-temporal variations in the fecundity of brown trout Salmo trutta L.: Trade-offs between egg size and number. Freshwater Biology. 38(2): 277-288
Maddern MG, Morgan DL, Gill HS. (2007). Distribution, diet and potential ecological impacts of the introduced Mozambique mouthbrooder Oreochromis mossambicus Peters (Pisces: Cichlidae) in Western Australia. Journal of the Royal Society of Western Australia. 90(4): 203-214
Manangkalangi E. (2009). Makanan, pertumbuhan dan reproduksi ikan pelangi arfak (Melanotaenia arfakensis Allen) di Sungai Nimbai dan Sungai Aimasi, Manokwari. [Tesis]. Bogor (ID): Institut Pertanian Bogor. 105 hal
Manangkalangi E, Rahardjo MF, Sjafii DS. (2009a). Habitat ontogeni ikan pelangi arfak (Melanotaenia arfakensis) di Sungai Nimbai dan Sungai Aimasi, Manokwari. Jurnal Natural. 8(1): 4-11
Manangkalangi E, Rahardjo MF, Sjafii DS, Sulistiono. 2009b. Musim pemijahan ikan pelangi arfak (Melanotaenia arfakensis Allen) di Sungai Nimbai dan Sungai Aimasi, Manokwari. Jurnal Iktiologi Indonesia. 9(1): 1-12
Manangkalangi E, Rahardjo MF, Sjafii DS, Sulistiono. (2009c). Pengaruh Kondisi Hidrologi Terhadap Komunitas Makroavertebrata di Sungai Aimasi dan Sungai Nimbai, Manokwari. Jurnal Perikanan dan Kelautan. 5(2): 99-110
Manangkalangi E, Leatemia SPO, Lefaan PT, Peday HFZ, Sembel L. (2014). Kondisi habitat ikan pelangi arfak, Melanotaenia arfakensis, 1990 di Sungai Nimbai, Prafi Manokwari. Jurnal Iktiologi Indonesia. 14(1): 21-36
Manangkalangi E, Rahardjo MF, Hadiaty RK, Hariyadi S. (2017). Efektivitas ikan pelangi arfak, Melanotaenia arfakensis Allen, 1990 dalam mencari makan pada tingkat kekeruhan air yang berbeda: suatu pendekatan laboratorium. Jurnal Iktiologi Indonesia. 17(3): 299-310
Manangkalangi E, Rahardjo MF, Hadiaty RK, Hariyadi S, Simanjuntak CPH. (2019). Trophic ecology of fish community at Nimbai Stream: competition and predation interaction to Arfak rainbowfish, Melanotaenia arfakensis Allen, 1990. Jurnal Iktiologi Indonesia. 19(3): 449-464
Manangkalangi E, Rahardjo MF, Hadiaty RK, Hariyadi S, Simanjuntak CPH. (2020). Distribution and abundance of the Arfak rainbowfish, Melanotaenia arfakensis Allen, 1990 in Prafi River system, Manokwari, West Papua: due to habitat degradation? IOP Conference Series: Earth and Environmental Science. 404: 012043
Miller TJ, Crowder LB, Rice JA, Marschall EA. (1988). Larval size and recruitment mechanisms in fishes: toward a conceptual framework. Canadian Journal of Fisheries and Aquatic Sciences. 45(9): 1657-1670
Milton DA, Arthington AH. (1984). Reproductive strategy and growth of the crimson-spotted rainbowfish, Melanotaenia splendida fluviatilis (Castelnau) (Pisces: Melanotaeniidae) in south-eastern Queensland. Australian Journal of Marine and Freshwater Research. 35(1): 75-83
Morgan DL, Gill HS, Maddern MG, Beatty SJ. (2004). Distribution and impacts of introduced freshwater fishes in Western Australia. New Zealand Journal of Marine and Freshwater Research. 38(3): 511-523
Olden JD, Kennard MJ. (2010). Intercontinental comparison of fish life history strategies along a gradient of hydrologic variability. In: Gido KB, Jackson DA (eds.). Community ecology of stream fishes: concepts, approaches, and techniques. Proceedings of a symposium held in Ottawa, Ontario, Canada, 19-20 August 2008. Bethesda (USA): American Fisheries Society. pp. 83-107
Oliveira RF, Almada VC. (1998). Maternal aggression during the mouthbrooding cycle in the cichlid fish, Oreochromis mossambicus. Aggressive Behavior. 24: 187-196
Oliveira RF, Almada VC, Canário AVM. (1996). Social modulation of sex steroid concentrations in the urine of male cichlid fish Oreochromis mossambicus. Hormones and Behavior. 30(1): 2-12
Olivotto I, Holt SA, Carnevali O, Holt GJ. (2006). Spawning, early development, and first feeding in the lemonpeel angelfish Centropyge flavissimus. Aquaculture. 253(1-4): 270-278
Oulton LJ, Haviland V, Brown C. (2013a). Predator recognition in rainbowfish, Melanotaenia duboulayi, embryos. PLoS ONE. 8(10): e76061
Oulton LJ, Carbia P, Brown C. (2013b). Hatching success of rainbowfish eggs following exposure to air. Australian Journal of Zoology. 61(5): 395-398
Philippart JC. (1992). Is captive breeding an effective solution for the conservation of endemic species? Biological Conservation. 72(2): 281-295
Portella MC, Dabrowski K. (2008). Diets, physiology, biochemistry and digestive tract development of freshwater fish larvae. In: Cyrino JEP, Bureau DP, Kapoor BG (eds.). Feeding and digestive functions in fishes. Enfield, NH (USA): Science Publishers. pp. 227-279
Pusey BJ, Arthington AH, Bird Jr JR, Close PG. (2001). Reproduction in three species of rainbowfish (Melanotaeniidae) from rainforest streams in northern Queensland, Australia. Ecology of Freshwater Fish. 10(2): 75-87
Pusey BJ, Arthington AH, Close PG, Bird JR. (2002). Larval fishes in rainforest streams: recruitment and microhabitat use. Proceedings of the Royal of Queensland. 110: 27-46
Radael MC, Cardoso LD, Andrade DR, Mattos D, Motta JH, Manhães JV, Vidal Jr MV. (2013). Morphophysiological characterization of the embryonic development of Melanotaenia praecox (Weber & de Beaufort, 1922). Zygote. 22(4): 533-539
Radael MC, Cardoso LD, de Andrade DR, Ferreira AV, de Cruz Mattos D, Vidal Jr MV. (2015). Effect of temperature on embryonic development of Melanotaenia boesemani (Allen and Cross, 1982). Zygote. 24(2): 301-309
Reid HP, Holdway DA. (1995). Early development of the Australian crimson-spotted rainbowfish, Melanotaenia fluviatilis (Pisces: Melanotaeniidae). Marine and Freshwater Research. 46(2): 475-480
Rice JA, Crowder LB, Holey ME. (1987). Exploration of mechanisms regulating larval survival in Lake Michigan bloater: a recruitment analysis based on characteristics of individual larvae. Transactions of the American Fisheries Society. 116(5): 703-718
Rønnestad I, Morais S. (2008). Digestion. In: Finn RN, Kapoor BG (eds.). Fish larval physiology. Enfield, NH (USA): Science Publishers. pp. 201-262.
Schönhuth S, Luikart G, Doadrio I. (2003). Effects of a founder event and supplementary introductions on genetic variation in a captive breeding population of the endangered Spanish killifish. Journal of Fish Biology. 63(6): 1538-1551
Siby LS, Rahardjo MF, Sjafei DS. 2009. Biologi reproduksi ikan pelangi merah (Glossolepis incisus, Weber 1907) di Danau Sentani. Jurnal Iktiologi Indonesia. 9(1): 49-61
Simanjuntak, C.P.H., Kinoshita I, Fujita S, Takeuchi K. (2015). Reproduction of the endemic engraulid, Coilia nasus, in freshwaters inside a reclamation dike of Ariake Bay, western Japan. Ichthyological Research. 62 (3): 374-378
Singh RK, Khandagale PA, Chavan SL, Sapkale PH. (2006). The relationship of ova diameter to fertilization rates, hatching rates, survival percentages and specific growth rates in the common carp and indian major carps. Asian Fisheries Society. 19(3): 257-269
Snyder NFR, Derrickson SR, Beissinger SR, Wiley JW, Smith TB, Toone WD, Miller B. (1996). Limitations of captive breeding in endangered species recovery. Conservation Biology. 10(2): 338-348
Winemiller KO, Rose KA. (1992). Patterns of life-history diversification in North American fishes: implications for population regulation. Canadian Journal of Fisheries and Aquatic Sciences. 49(10): 2196-2218.
Wootton RJ. (1984). Introduction: tactics and strategies in fish reproduction. In: Potts GW, Wootton RJ (eds.). Fish reproduction: strategies and tactics. New York (USA): Academic Press. pp. 1-12.
Wootton RJ. (1990). Ecology of Teleost Fishes. New York (USA): Chapman and Hall Ltd. 404 p
Yúfera M, Darias MJ. (2007). The onset of exogenous feeding in marine fish larvae. Aquaculture. 268(1-4): 53-63.
Yuliani F, Musthofa SZ, Kadarini T, Elfidasari D. (2013). Perkembangan larva ikan rainbow boesemani (Melanotaenia boesemani): tahap pembentukan sirip dan pembelokan tulang ekor. Unnes Journal of Life Science. 2(2): 100-104.
Zambonino-Infante JL, Gisbert E, Sarasquete C, Navarro I, Gutiérrez J, Cahu CL. (2008). Ontogeny and physiology of the digestive system of marine fish larvae. In: Cyrino JEP, Bureau DP, Kapoor BG (eds.). Feeding and digestive functions in fishes. Enfield, NH (US).